
How to find the number of Protons, Neutrons and Electrons? Chemistry YouTube
Primarily, the atomic structure of matter is made up of protons, electrons and neutrons. The protons and neutrons make up the nucleus of the atom, which is surrounded by the electrons belonging to the atom. The atomic number of an element describes the total number of protons in its nucleus. Neutral atoms have equal numbers of protons and.

Neutron Discovery, Difference and more Teachoo Concepts
So yeah, " carbon " has six protons, six neutrons and six electrons. Carbon-12 specifically, which accounts for 98.9 percent of all carbon. The chemical formula for melanin is C 18 H 10 N 2 O 4. That means there are 18 carbon atoms, 10 hydrogen, 2 nitrogen and 4 oxygen atoms in a melanin molecule. Melanin is carbon, hydrogen, nitrogen and.

Electrons — Structure & Properties Expii
Because the sum of the numbers of protons and neutrons equals the mass number, 127, the number of neutrons is 74 (127 − 53 = 74). Since the iodine is added as a 1− anion, the number of electrons is 54 [53 - (1-) = 54]. Exercise. An ion of platinum has a mass number of 195 and contains 74 electrons.

Het aantal neutronen, protonen en elektronen bepalen wikiHow
It has an atomic number of 6. That means a carbon atom has 6 protons, 6 neutrons, and 6 electrons. Since carbon is in the second row (or second period), it has 2 electron orbits. Use the clay to make your protons and neutrons in the nucleus. The nucleus should be about the size of a large gumball. (Use two different colors of clay.)

Nucleons, Atomic Number and Mass Number Definitions.. and more
Lithium has 3 protons, 4 neutrons and 3 electrons. 4. Beryllium has 4 protons, 5 neutrons and 4 electrons. 5. Boron has 5 protons, 6 neutrons and 5 electrons. 6.

Periodic Table Electrons And Protons Periodic Table Timeline 1EE
Study with Quizlet and memorize flashcards containing terms like What is the atomic number of an atom that has 6 protons, 6 neutrons, and 6 electrons? 12 0 18 -1 6, Which of these refers to atoms with the same atomic number but different atomic masses? These atoms have different numbers of protons. These atoms have different numbers of electrons. These atoms are isomers. These atoms are.
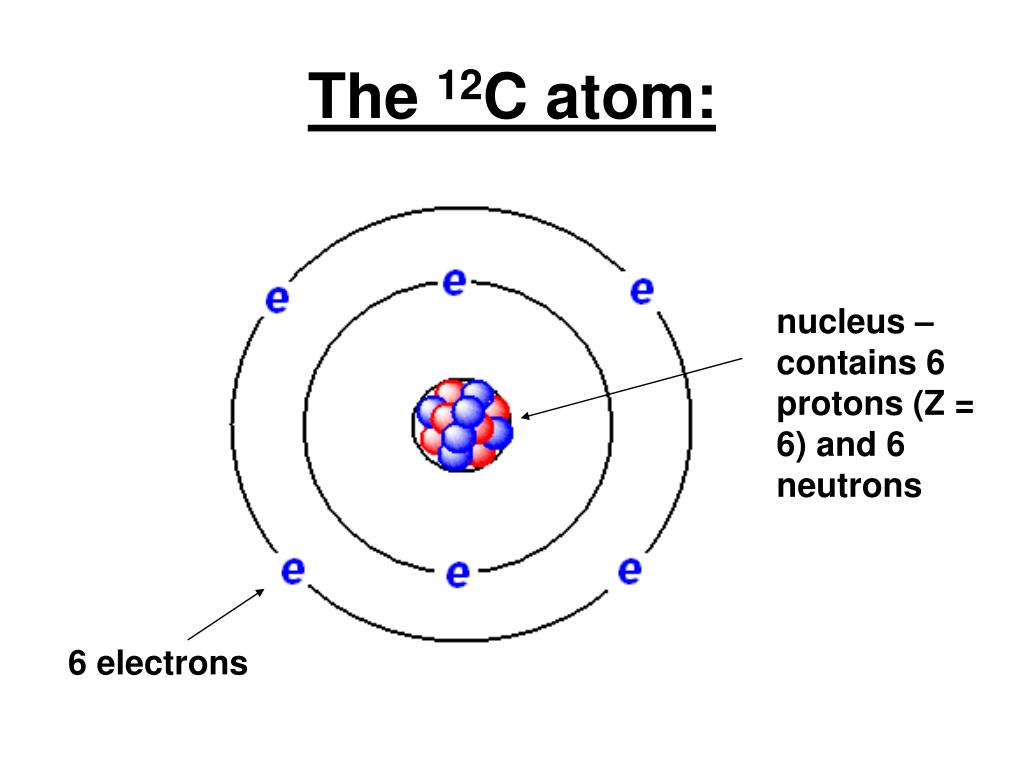
PPT Lecture 4. Chapter 2. Structure of the Atom (Contd.) PowerPoint Presentation ID3828875
Together, the number of protons and the number of neutrons determine an element's mass number: mass number = protons + neutrons. If you want to calculate how many neutrons an atom has, you can simply subtract the number of protons, or atomic number, from the mass number. A property closely related to an atom's mass number is its atomic mass.
Periodic Table Of Elements List With Protons Neutrons And Electrons Awesome Home
Resources. Education. Graphite 101. Carbon by the Number. Carbon is a chemical element. Its atomic number is 6; its atomic weight is 12.011. It is a group IVA element, residing between boron and nitrogen on the periodic table, and it has 6 protons, 6 neutrons, and 6 electrons. The electron configuration is 1s2 , 2s2, 2p2.

Periodic Table Carbon Protons Neutrons Electrons Periodic Table Timeline
Explanation: atomic number =number of protons. Therefore it is 6. Answer link. The atomic number is 6, for carbon. The atomic number is equal to the number of protons.
.PNG)
Periodic Table Of Elements Number Of Protons Neutrons And Electrons About Elements
Each atom has a charged sub-structure consisting of a nucleus, which is made of protons and neutrons, surrounded by electrons. The number of protons and the mass number of an atom define the type of atom. Atoms of the same element with different mass numbers are called isotopes. Created by Jay.
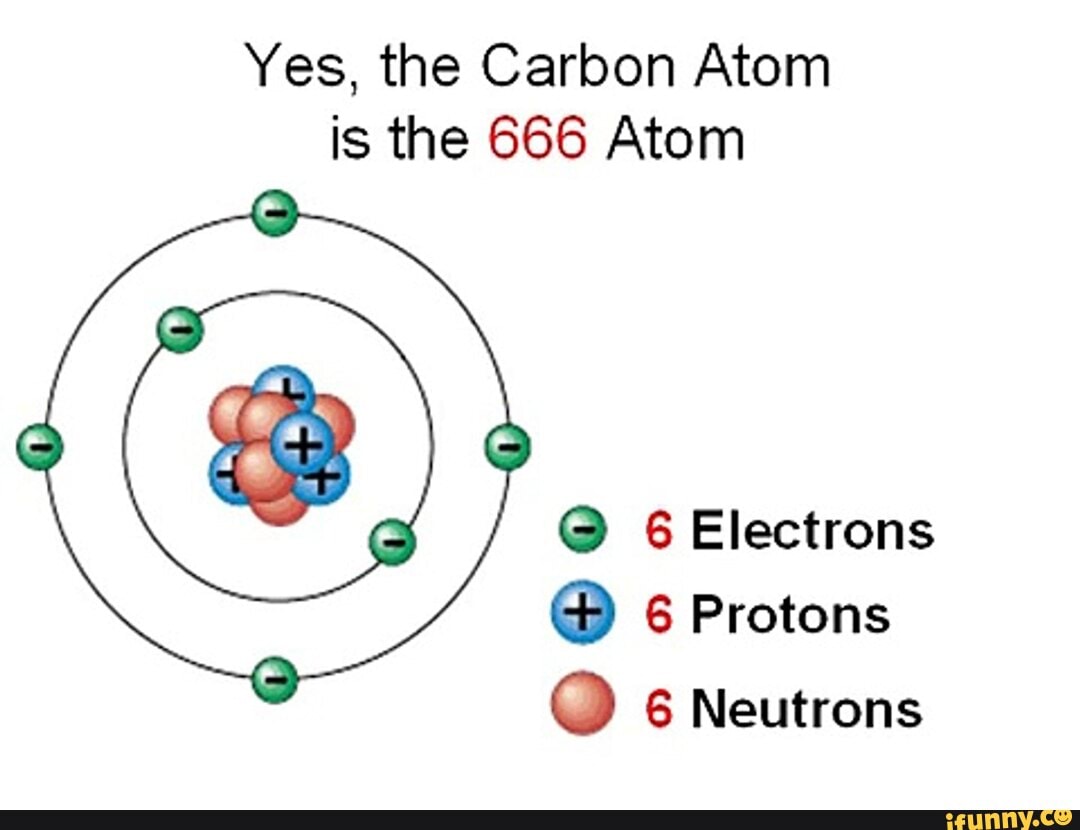
Yes, the Carbon Atom is the 666 Atom 6 Electrons 6 Protons 6 Neutrons iFunny
Protons and Neutrons in Carbon. Carbon is a chemical element with atomic number 6 which means there are 6 protons in its nucleus. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z.The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10-19 coulombs.

Atoms & Molecules echapter — The Biology Primer
The reduced graphene oxide connection with 3, 4 and 5G and the higher densities from graphene domes or death towers (1000 domes or towers minimum are now operational in every city around the World) are sourced with lasers that have been stripped of their regular electromagnetic waves, leaving only high density magnetic quantum percolation capable of atom to atom interconnects.
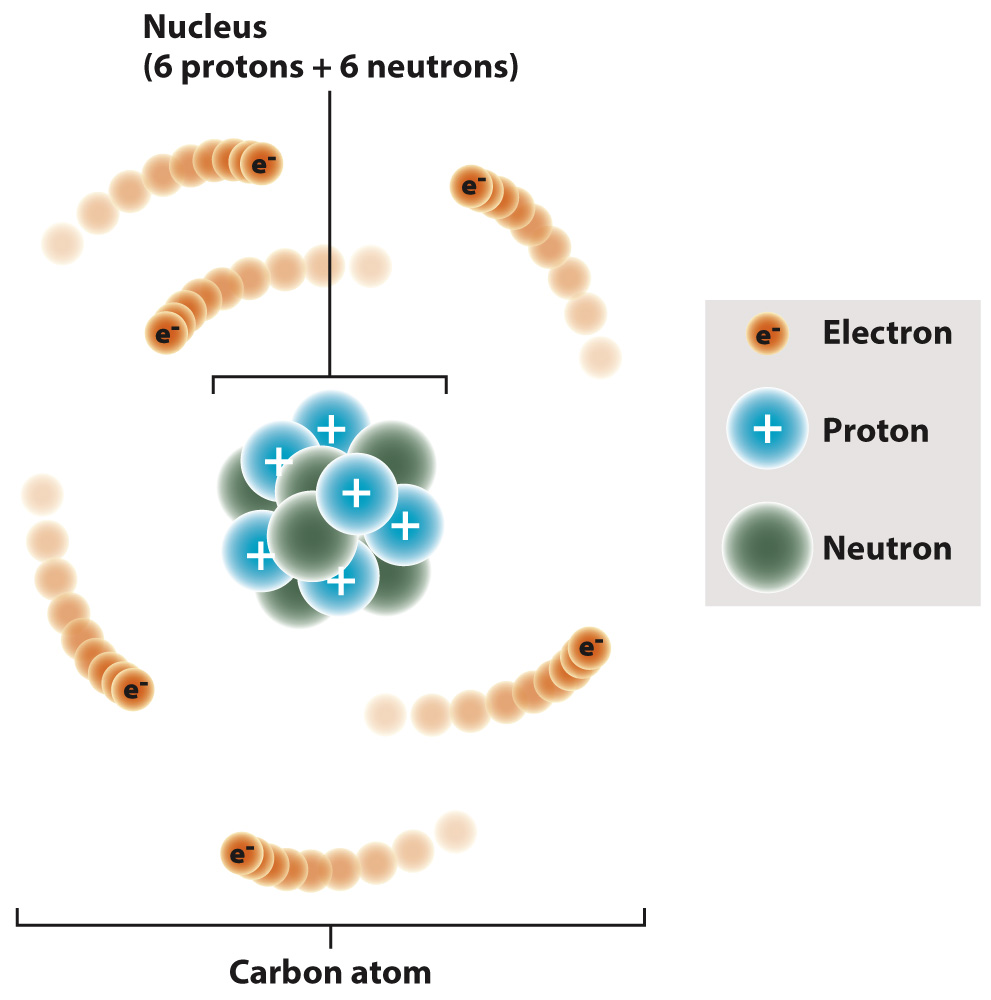
image
Carbon is a chemical element. Its atomic number is 6; its atomic weight is 12.011. It is a group IVA element, residing between boron and nitrogen on the periodic table, and it has 6 protons, 6 neutrons, and 6 electrons.
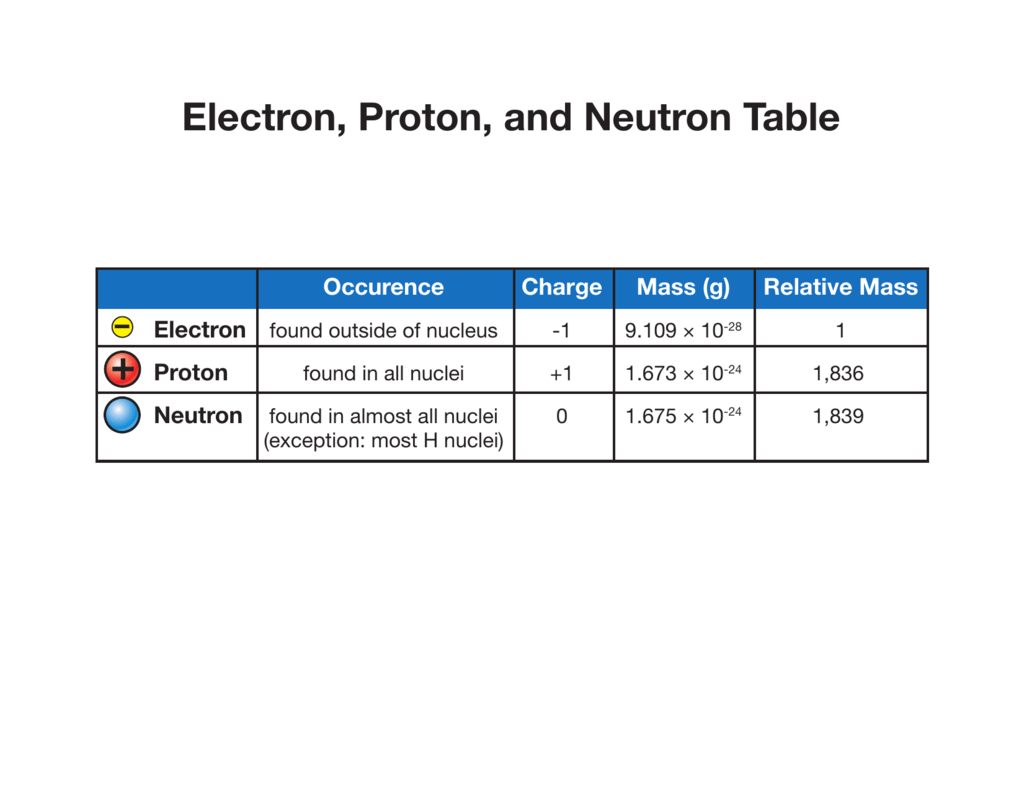
Electron, Proton, and Neutron Table
Study with Quizlet and memorize flashcards containing terms like An atom has 6 protons, 8 neutrons, and 6 electrons. Its atomic mass is:, An atom with 6 protons, 7 neutrons, and 6 electrons shares four pairs of electrons with four other atoms. This atom is now considered to be :, Nucleotides are composed of and more.

Protons neutrons and subatomic particles Pharmacy Gyan
While protons and neutrons are located inside the nucleus at the center of the atom, electrons are located outside the nucleus in what is often called the electron cloud. Figure 4.4.1 4.4. 1: Electrons are much smaller than protons or neutrons. If an electron was the mass of a penny, a proton or a neutron would have the mass of a large bowling.
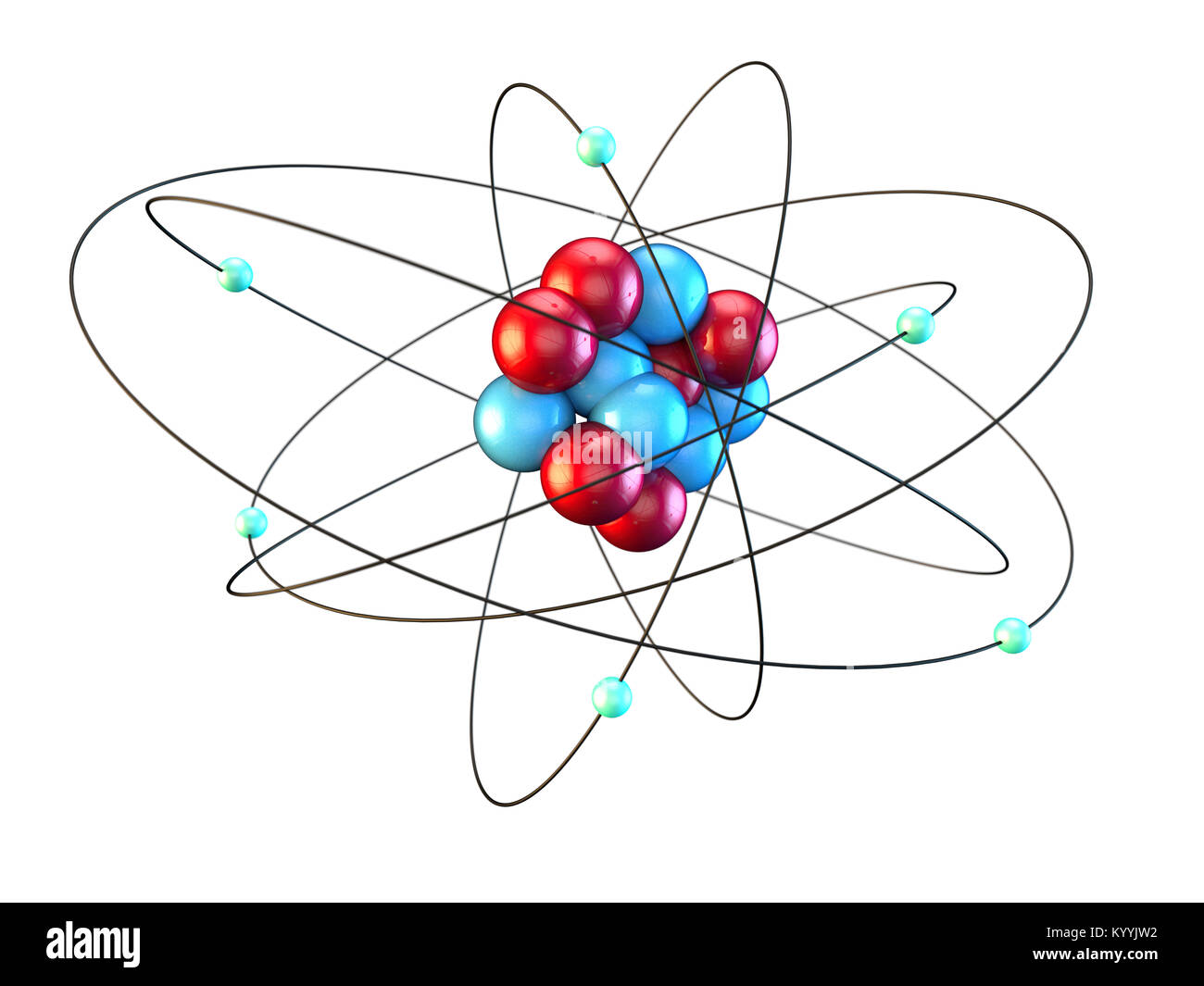
Carbon atom showing six electrons orbiting six protons and six neutrons, chemistry chemical
Study with Quizlet and memorize flashcards containing terms like What is the atomic number of an atom that has 6 protons, 6 neutrons, and 6 electrons? 12 6 18 0 -1, Which of these refers to atoms with the same atomic number but different atomic masses? These atoms have different numbers of protons. These atoms have different numbers of electrons. These atoms are different elements. These atoms.